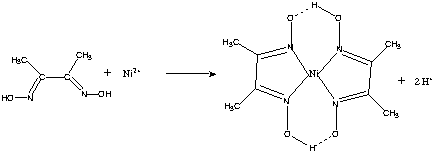 
Finally, mass spectrometry analysis revealed that DMG can coordinate Cu or Ni, but not Fe, Se or Zn. Addition of the Ni-specific chelator dimethylglyoxime (DMG) inhibited Aβ 40 aggregation in absence of added metal, as well as in presence of Cu 2+ and Ni 2+, but not in presence of Zn 2+. Nickel binding to Aβ 40 peptide was confirmed by isothermal titration calorimetry. Using a fluorescence-based assay, we found that Aβ 40 peptide in vitro aggregation was enhanced by addition of Zn 2+ and Ni 2+, and Ni 2+-induced aggregation was facilitated by acidic conditions. Cu and Zn were among the metals detected unexpectedly, nickel (Ni) was one of the most abundant elements. In the present study, inductively coupled plasma mass spectrometry (ICP-MS) was used to determine the metal content of a commercial recombinant human Aβ 40 peptide. Previous studies have shown that select metal ions, most specifically copper (Cu) and zinc (Zn) ions, have a synergistic effect on the aggregation of Aβ-peptides. One of the hallmarks of the most common neurodegenerative disease, Alzheimer’s disease (AD), is the extracellular deposition and aggregation of Amyloid Beta (Aβ)-peptides in the brain.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
